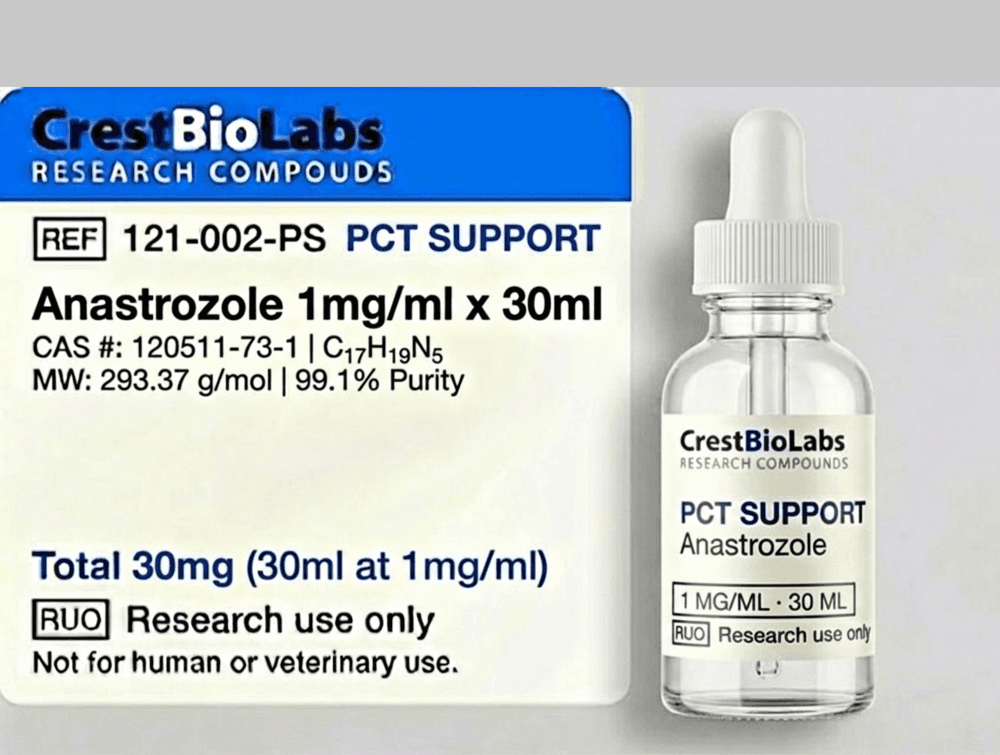
Anastrozole 1mg x 30ml
Anastrozole is a potent and selective non-steroidal aromatase inhibitor used in research settings.
Quick Reference
This product is temporarily unavailable. Join our notification list to be the first to know when it's back in stock.
About Anastrozole 1mg x 30ml
Anastrozole is a third-generation aromatase inhibitor that selectively inhibits the enzyme aromatase, which is responsible for converting androgens to estrogens. It is a widely researched compound in endocrine studies. This product is supplied as a research solution at 1mg/ml concentration.
Mechanism of Action
Selectively and reversibly binds the heme group of cytochrome P450 aromatase (CYP19A1), inhibiting the conversion of androgens (testosterone, androstenedione) to estrogens (estradiol, estrone) with >99% suppression of plasma estradiol at standard research concentrations.
Research Applications
Anastrozole, a potent and selective aromatase inhibitor, ver...
Journal of Clinical Oncology (1997)
A randomised trial of anastrozole (Arimidex) versus tamoxife...
European Journal of Cancer (2000)
Pharmacokinetics and pharmacodynamics of anastrozole
Expert Opinion on Drug Metabolism & Toxicology (2005)
Product Quick Facts
HPLC Verified
99.1% purity confirmed
COA Available
Full chromatograms & MS data
Same-Day Shipping
Orders before 2 PM EST
Cold Chain
Temperature-controlled packaging
Handling Protocol
- •Store at room temperature, protect from light
- •Reconstitute with bacteriostatic water
- •Avoid repeated freeze-thaw cycles
- •Handle under sterile conditions
Frequently Asked Questions About Anastrozole 1mg x 30ml
All three are aromatase inhibitors but differ in mechanism, potency, and reversibility: Anastrozole and Letrozole are non-steroidal, reversible, competitive inhibitors that bind the heme iron of CYP19A1, while Exemestane is a steroidal, irreversible (suicide) inhibitor that permanently inactivates the enzyme by covalent binding to the substrate pocket. Letrozole is approximately 10-20-fold more potent than Anastrozole in cell-free aromatase assays (IC50 ~11.5 nM vs. ~15 nM), though both achieve >95% estradiol suppression at standard research concentrations. Exemestane's irreversible mechanism means aromatase activity only recovers via de novo enzyme synthesis (half-life of recovery ~2-3 days), whereas Anastrozole's inhibition reverses as the compound clears, making it preferable for studies requiring precise temporal control of estrogen suppression windows.
Reversible inhibition means Anastrozole non-covalently occupies the active site of CYP19A1 aromatase through competitive binding to the heme iron coordination site, and once Anastrozole concentrations decline through metabolism or clearance, the enzyme regains full catalytic activity. This contrasts with Exemestane, which forms an irreversible covalent bond with the enzyme active site, permanently destroying that enzyme molecule's function. For research design, reversibility enables washout experiments where investigators can suppress estradiol for a defined period, then withdraw Anastrozole and monitor the kinetics of estrogen recovery — a protocol impossible with irreversible inhibitors without waiting for complete enzyme re-synthesis. The 40-50-hour half-life means approximately 7-10 days (5-6 half-lives) are needed for full washout and restoration of baseline aromatase activity.
Unlike lyophilized peptides that require reconstitution with bacteriostatic or sterile water, this Anastrozole product is a ready-to-use 1 mg/mL solution in 30 mL format that requires no reconstitution — simply agitate gently before each use to ensure homogeneity. The liquid formulation uses an organic solvent system (Anastrozole is sparingly soluble in water but freely soluble in methanol, ethanol, and DMSO), so researchers should verify solvent compatibility with their specific assay system. Storage is at room temperature protected from light, which is a significant departure from the -20°C requirement for most peptides. Researchers should note the solution concentration when calculating volumetric doses: at 1 mg/mL, standard micropipettes can deliver precise microgram-level quantities without the dilution errors common when working with sub-milligram powder weighing.
Anastrozole in solution is chemically stable for 12-24 months at room temperature when protected from light and stored in an airtight container, owing to the compound's robust triazole ring system and lack of easily oxidizable functional groups. Degradation products typically arise from photolytic cleavage of the benzyl-triazole bond when exposed to UV light, which is why amber or opaque storage is recommended. Visible signs of degradation include color change from clear to yellow or the formation of precipitate, either of which should prompt researchers to discard the solution. Analytical verification by HPLC or LC-MS can confirm potency if there is any doubt — the parent compound elutes with a characteristic retention time that shifts or splits when significant degradation has occurred.
Anastrozole's selectivity arises from its triazole nitrogen atoms coordinating specifically with the heme iron of CYP19A1, combined with the flanking cyanopropyl groups that occupy the unique hydrophobic substrate channel of the aromatase active site — a channel geometry distinct from other CYP450 family members. At pharmacologically relevant concentrations, Anastrozole shows minimal inhibition of CYP1A2, CYP2C8/9, CYP2D6, or CYP3A4, which is important for researchers conducting multi-compound studies where CYP-mediated interactions could confound results. This enzyme selectivity was demonstrated through comparative IC50 profiling across CYP450 panels, where Anastrozole's IC50 for CYP19A1 was orders of magnitude lower than for any other CYP enzyme tested.
The MCF-7 aromatase-transfected cell line (MCF-7aro) is the most widely used in vitro model, as it expresses high levels of CYP19A1 and converts androstenedione to estradiol in a quantifiable, dose-dependent manner that Anastrozole inhibits with a clear IC50 curve. For in vivo work, the ovariectomized rodent supplemented with exogenous androgen substrate provides a clean system where all measurable estradiol is aromatase-derived, eliminating ovarian estrogen as a confound. Human placental microsome preparations offer a cell-free system for direct enzyme kinetics studies, enabling Ki determination and Lineweaver-Burk analysis of Anastrozole's competitive inhibition mechanism. More recently, patient-derived xenograft models have been used to study aromatase inhibition in a translational research context.
The extended half-life means Anastrozole reaches steady-state concentrations after approximately 7-10 days of repeated administration, which researchers must account for when designing chronic suppression studies — measuring estradiol before steady state is achieved will underestimate the compound's maximal suppressive effect. For acute studies, the long half-life also means that a single administration produces a sustained pharmacodynamic effect lasting 3-5 days, reducing the need for frequent redosing and minimizing handling stress in animal models. This pharmacokinetic profile creates a wide and forgiving dosing window, but it also means washout periods must be extended (minimum 10-14 days) when researchers need to restore baseline estrogen levels between experimental phases.
Anastrozole's 1,2,4-triazole ring donates a lone electron pair from its N-4 nitrogen to the ferric (Fe3+) heme iron at the active site of CYP19A1, forming a coordinate bond that displaces the molecular oxygen normally required for the aromatization reaction of androgens to estrogens. This coordination prevents the three sequential hydroxylation steps that CYP19A1 performs on the C19 methyl group of androstenedione during its conversion to estrone. The two 2-cyanopropan-2-yl groups flanking the central phenyl ring provide steric complementarity with the hydrophobic substrate access channel, contributing binding affinity (Kd in the low nanomolar range) while excluding the compound from the differently shaped channels of other CYP450 isoforms. Because this is a coordinate bond rather than a covalent modification, enzyme activity is fully restored upon Anastrozole dissociation, which is the structural basis for its classification as a reversible inhibitor.
You May Also Need
Bacteriostatic Water (BAC) 3ml
H₂O + 0.9% C₇H₈O (benzyl alcohol)
Bacteriostatic Water (BAC) is USP-grade sterile water containing 0.9% benzyl alcohol as a bacteriostatic preservative. It is the standard reconstitution solvent for lyophilized research peptides.
